UNC’s Ronit Freeman is working to understand how Omicron’s mutations make it the most infectious variant yet.

Omicron and its sublineages remain a major public health concern owing to their high infectivity and antibody evasion. Perhaps the most challenging is that the sensitivity of some of the available rapid tests have been reduced against Omicron, compared to previous variants, forcing people to rethink COVID-19 testing, according to new research.
Freeman has previously developed sugar-labeled test strips termed GlycoGrip, leveraging the interactions SARS-CoV-2 has with cell surface sugars to reliably detect it.
Freeman’s approach to technology and innovation takes guidance from nature. “If we are able to understand the consequences of new mutations on how the virus interacts with the sugar coat of cells, we might be able to optimize those interactions on our test strips,” said Freeman.
Now, Freeman, in collaboration with researchers from UCSD, examined how the increased positive charge on the surface of spike proteins of variants is affecting the virus’ interactions with its receptors.
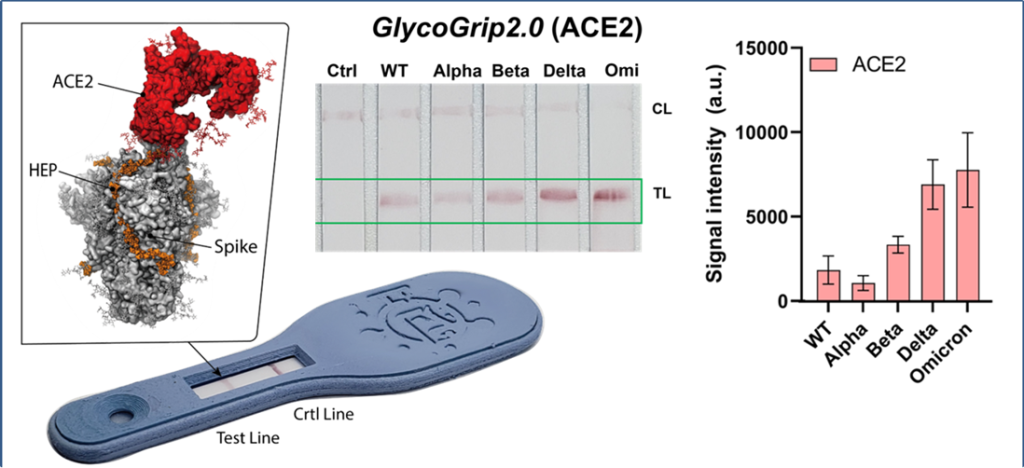 The consequence of change in surface charge is dramatic, causing the virus to be strongly pulled towards the sugar-rich negatively charged cell surface. Not only does Omicron bind these sugars stronger, but it also uses them to synergistically enhance its interactions with its primary receptor, ACE-2.
The consequence of change in surface charge is dramatic, causing the virus to be strongly pulled towards the sugar-rich negatively charged cell surface. Not only does Omicron bind these sugars stronger, but it also uses them to synergistically enhance its interactions with its primary receptor, ACE-2.
“This ‘fatal’ attraction is one of the reasons Omicron is so infectious,” said Freeman.
Freeman and her team were able to convert the discovery of a tertiary complex between the Omicron spike proteins, cell surface sugars, and ACE-2, into a highly reliable sensor, that not only detects Omicron, but picks it up better than previous variants.
The findings were published April. 7 in the journal Cell Reports Physical Science.
“Our detection technology co-evolves with the virus,” said Freeman.
By leveraging the virus’s evolving candy crush, GlycoGrip is effective in testing for all current variants and is expected to capture future variants as well. The team is now working to commercialize GlycoGrip, as well as expand the technology for the detection of a wide array of viruses.
Ronit Freeman is an associate professor in the department of applied physical sciences in the College of Arts and Sciences.
The paper is titled “SARS-CoV-2 evolved variants optimize binding to cellular glycocalyx.” The full author list includes: Sanghoon Kim, Fiona Kearns, Mia Rosenfeld, Lane Votapka, Lorenzo Casalino, Micah Papanikolas, Rommie E. Amaro and Ronit Freeman.
The research was funded by Research Corporation for Science Advancement (COVID Initiative grant #27350) award, the North Carolina Policy Collaboratory at the University of North Carolina at Chapel Hill with funding from the North Carolina Coronavirus Relief Fund established and appropriated by the North Carolina General Assembly, NSF RAPID (DMS-2028758, MCB-2032054), NIH GM132826, UC San Diego Moores Cancer Center 2020 SARS-COV-2 seed grant, U19-AI171954 from NIAID.
